Toxicity measurements with Aliivibrio fischeri (formerly Vibrio fischeri) with a plate reader. Operator saves up to 90 % of hands on time with the dedicated luminometer & software compared to competitor’s method! You can prepare 6 sample dilutions in duplicate faster than you put the test tubes into a rack!
ISO STANDARD 21338 method is based on this measurement! Read more from below.
Automated measurement of 6 samples in duplicates, automated result calculation. Save time, save money. Automated reagent priming, automated washing.
Bioluminescent Vibrio fischeri has been widely used for measuring the acute toxicity of water samples and chemical substances. The test is standardised (EN ISO 11348) and there is several commercial test systems available (Microtox, BioTox, LumiStox, ToxAlert etc.). The sample is incubated in contact with the bacteria for 15 or 30 minutes and the luminescence intensity after the incubation is compared to the luminescence intensity of pure bacteria. Reduction in light production is regarded as toxicity. The method is rapid and it is known to be especially sensitive for organic contaminants. The main disadvantage is that colour and turbidity present in most of the natural samples scatter the light and are being seen as toxicity. The methods for correcting the effects of colour and turbidity are time consuming, inconvenient and somewhat unreliable.
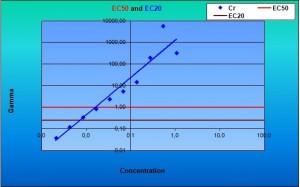
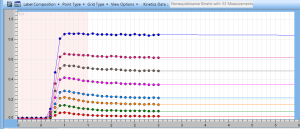
The Flash test is an improved BioToxTM test, originally designed for solid samples. The method takes account the colour and turbidity of the sample during the whole measurement. The measurement is accomplished in kinetic mode with automated luminometer capable of dispensing and measuring at the same time. In the Flash method the V. fischeri bacteria are dispensed into the sample and the signal is recorded continuously for 30 seconds. The maximum signal received immediately after dispensing (Ipeak) is compared to the signal after 30 seconds incubation period (I30s). With most of the chemicals, especially organic ones, the toxic effect (reduction in light production) can be seen already after few seconds of incubation. However, the effect of some chemicals can be seen only after longer contact times. Thus, kinetic data from samples after 15 or 30 min gives an additional dimension for obtaining reliable results.
The features described above makes the colour/turbidity correction methods unnecessary and facilitates the toxicity screening of majority of the samples within few seconds. The sensitivity of the Flash test with 30 s contact time is slightly reduced when compared to the standard method using 15 or 30 minutes contact times. When same contact times are used, the sensitivities of standard and Flash-methods are of same order of magnitude. The coefficient of variation is excellent being normally below 1%. Sample volume needed for one test is less than 1 ml and the capacity of the method is 40 to 80 samples/hour depending on the contact time used. NEW! Software and calculation for a PLATE READER LUMINOMETER!
Colour and turbidity are automatically corrected during the measurement. The results are generated automatically.
1243-500 BioTox kit Instructions for use solid diluent
Introduction
BioTox method is the traditional and standardized way to measure the toxicity of chemicals or effluents by utilising photobacteria (ISO 11348-3: Water quality – Determination of the inhibitory effect of water samples on the light emission of Vibrio fischeri (luminescent bacteria test))
The test is based on the fact that the light output of the bacteria is reduced, when it is introduced to toxic chemicals. The method is very rapid, it takes from 5 to 30 minutes to perform the test. The results with this system are comparable to published results with other photobacteria tests like Microtox® test.
The principle of the standard photobacteria measurement is that the bacteria and the sample are mixed together and after an incubation period the light output is measured.
BioTox luminometric toxity test is an ISO standardized (11348-3) acute toxicity test utilizing the photobacteria Aliivibrio fischeri (formerly Vibrio fischeri).
BioTox LumoPlate is a modified test for solid and colored samples according to the ISO Standard 21338. The BioTox ssystem include luminometer, computer program, reagent cooler / incubator and the test organism in freeze dried format. See below for more info.
Separate vials, buffers etc. available upon request.
Byonoy luminometer. More information to come shortly!
Titertek Berthodl FB14 for laboratory use (Standard ISO 11348-3 tests) and for field use. Measurements with FB14, Data handling and Toxicity estimation with Excel. Simple to use, cost effective system!
Berthod-Titertek Luminometer SiriusL for Standard ISO 21338 measurements.
BioTox system include the instrumentation and the reagents in a freeze dried form. The bacteria can be shipped to the customer at room temperature.
 |
Rapid – Cost-saving – Easy to use
|
Call or email us for futher info.